AI-Guided Ultrasound Elastography for Assessments of Chronic Liver Diseases
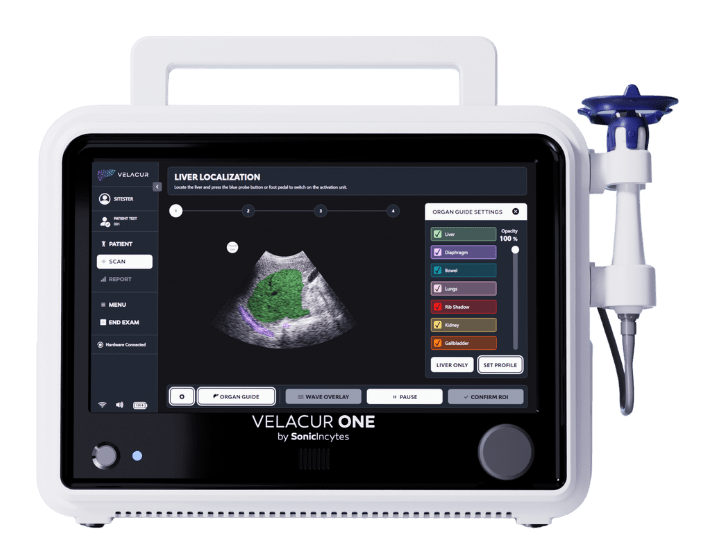

Velacur ONE is a point-of-care device that measures key indicators for MASLD and MASH, comparable to both invasive and non-invasive gold standards*.
*(ie. MRE/MRI-PDFF and biopsy)
Clinically validated against both invasive and non-invasive gold standards.
3-4X higher reimburesement compared to other non-imaging elastography.
*(ie. MRE/MRI-PDFF and biopsy)



Enables confident measurement, even with inexperienced operators.
Automatic wave detection to improve scan quality.
Enables confident measurement, even with inexperienced operators or high BMI patients.
Enables increased measurement depth and larger sample volume, with 2X the depth and 30X the volume of transient elastography.
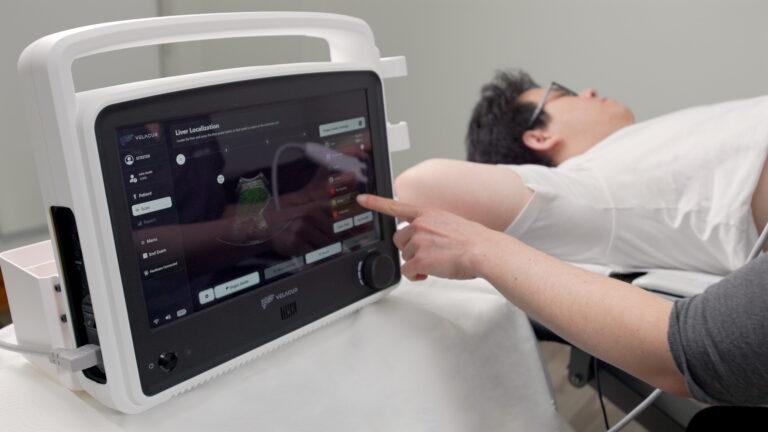
Our procedure is quick, comfortable and non-invasive and can be performed in-office by any trained medical professional.
Place the activation pad under the patient’s back to create and transmit multi-frequency steady state shear waves throughout the liver.
With AI guidance, operators use a sweeping motion to generate 3D volumetric measurements of liver tissue.
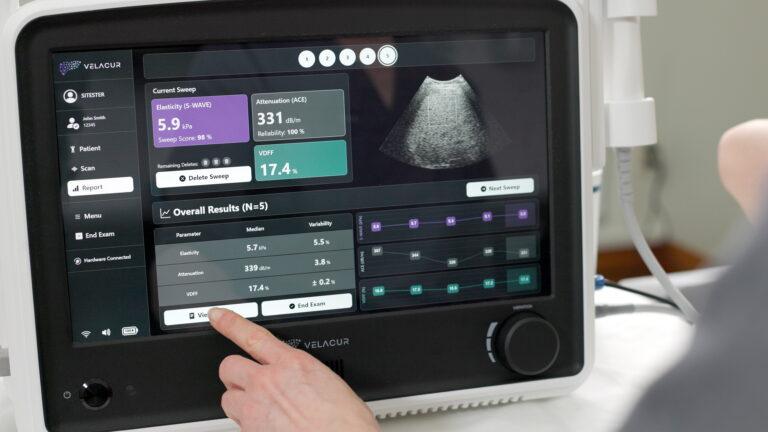
Elasticity and attenuation measurements are generated immediately allowing for patient consultation in the same visit.
Our procedure is quick, comfortable and non-invasive and can be performed in-office by any trained medical professional.
Place the activation pad under the patient’s back to create and transmit multi-frequency steady state shear waves throughout the liver.
With AI guidance, operators use a sweeping motion to generate 3D volumetric measurements of liver tissue.
Elasticity and attenuation measurements are generated immediately allowing for patient consultation in the same visit.
With real-time results, a low up-front cost and AI guidance, VELACUR makes liver imaging at the point of care affordable and accessible.
Easily locate the liver and go deeper to get measurements you’re confident in – even in technically challenging cases.